ABC Transporters-dependent Collateral Sensitivity
Observation - Earlier research had demonstrate that tumour cells that over-express ABCB1 (P-glycoprotein) are resistant to a large array of anti-cancer drugs, regardless of their structure and mechanisms of cytotoxicity. However, these same multi-drug resistant cells were hypersensitive or collaterally sensitive to other drugs that were NOT cytotoxic to normal cells or non-ABCB1 expressing tumour cells. These observations were initially made prior the characterization of ABCB1 drug interactions.
Decades later, 3rd generation drugs were developed that targeted ABCB1 and inhibited its drug efflux function, these drugs were termed "MDR reversing drugs or Chemosensitizers". However, while MDR1-reversing drugs are at best able to fully reverse ABCB1-mediated drug resistance, when used in combination with anti-cancer drugs (graph B; figure 1); collateral sensitivity drugs specifically target ABCB1-over-expressing tumour cells, when use ALONE in the absence of anticancer drugs.
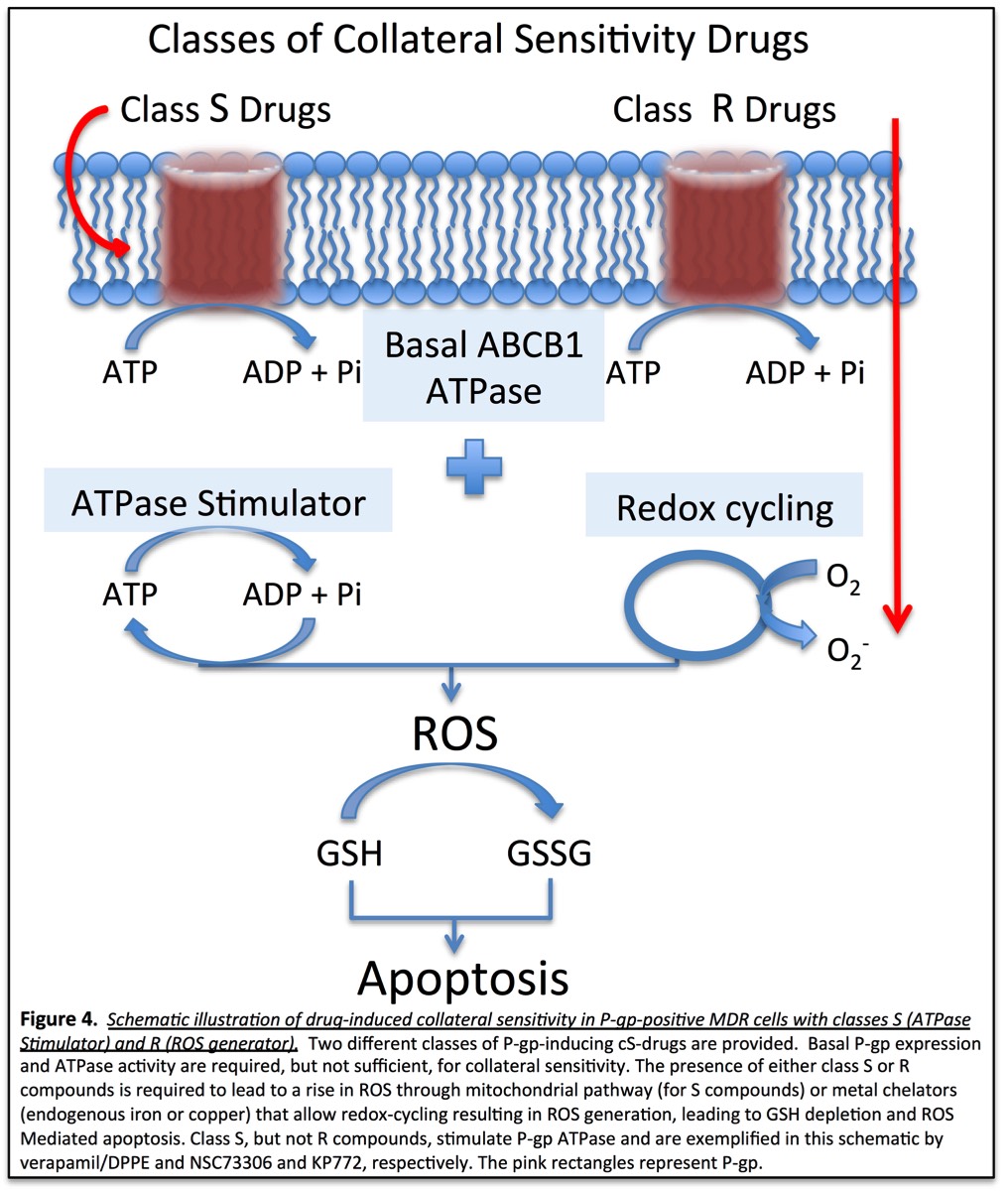
In an effort to resolve the molecular mechanism of ABCB1-dependent collateral sensitivity, Karwatsky et. al. (2003) showed that drugs that stimulated ABCB1-ATPase caused collateral sensitivity onto multi-drug resistant cells, while drugs that inhibited ABCB1-ATPase did NOT. These findings lead to our working hypothesis that stimulation of ABCB1-ATPase depleted cellular ATP levels and caused heightened oxidative stress in drug- resistant, but not in drug-sensitive cell (Figure 2).
In a later study, Laberge et. al. (2009) showed ABCB1 knock-down or inhibition of its ATPase activity reversed the collateral sensitivity phenotype, hence directly implicating ABCB1 expression or its ATPase in this phenotype. This mechanism has been confirmed with several drugs that activate ABCB1 ATPase activity (termed Class S drugs for ATPase Stimulating drugs). Alternatively, drugs that cause a sharp rise in reactive oxygen species through redox-cycling or inhibit complexes I or III of the mitochondria oxidative phosphorylation pathway also cause collateral sensitivity ONLY in the presence of high expression of ABCB1 (e.g., Class R drugs for Redox causative drugs; Figure 2).
A similar strategy was applied to drug resistant cells expressing another member of the ABC transporters, ABCC1 (or the multi drug resistant protein MRP1), which effluxes anti-cancer drugs and several normal cell metabolites (e.g., glutathione (GSH)). In that study, Laberge et. al. (2007) demonstrated that stimulating ABCC1 with the phytochemical apigenin causes a dramatic decrease in cellular GSH and heightened oxidative stress in drug resistant (ABCC1-expressing cells), but not in sensitive cells (ABCC1 negative cells).
Figure 1.
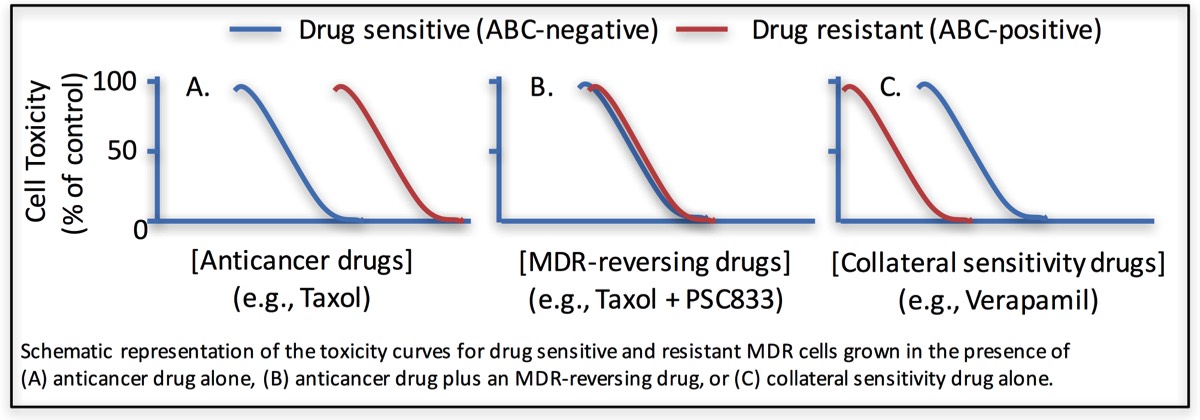
Figure 2
Our Hypothesis - Collateral Sensitivity - Is a phenotype observed in drug resistant tumour cells that over expression one of the ABC transporters. This phenotype is exposed when drug resistant cells are challenged with certain drugs. Tumour cells expressing different ABC transporter display collateral sensitivity via different mechanisms that collectively lead to oxidative stress-mediated cells death.
ABC-mediated collateral sensitivity can be activated with at least two classes of compounds: i) Class S drugs which stimulate ABC proteins ATPase activity and leads to dramatic increase in ATP level consumption, beyond basal levels, leading to heightened levels of reactive oxygen species (ROS) through mito-oxidative phosphorylation; or ii) Class R drugs which need not interact with ABC proteins but induce ROS in cells, that together with the basal levels of ABC-ATPase leads to enhanced ROS levels beyond a tolerable level causing apoptotic cell death. Class R type drugs lead to increased ROS through redox.
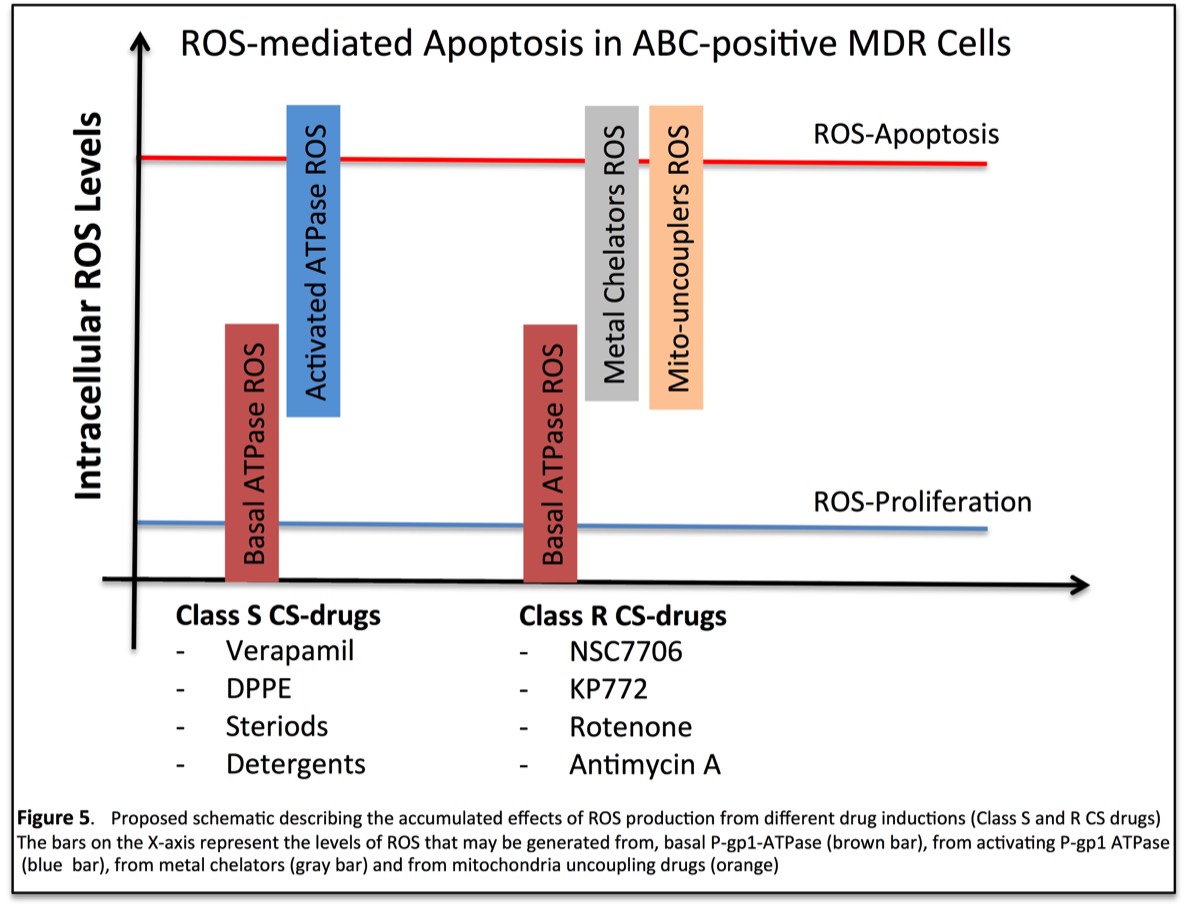
Figure 3
Overall Objective - It is well accepted that cancer cells will develop resistance to any and all drugs used in cancer chemotherapy, given exposure time and tumour cells' genomic instability. Moreover, tumour cells are likely to recruit more than one mechanism of resistance to chemotherapeutic drugs. However, earlier clinical studies have demonstrated over-expression of ABCB1 (or P-glycoprotein, P-gp) in patient samples, pre- and post-treatment, summarized in Figure 4 from Leonard, et. al. (2003). Given the percent of tumours that display increased P-glycoprotein (ABCB1) expression (40% - 85%), targeting P-glycoprotein-expressing tumour cells makes perfect sense. Unfortunately, earlier attempts to inhibit ABCB1 or P-glycoprotein function with "chemo-reversing or MDR-reversing drugs" were not successful due to unacceptable toxicity by interfering with its drug efflux function in normal tissues.
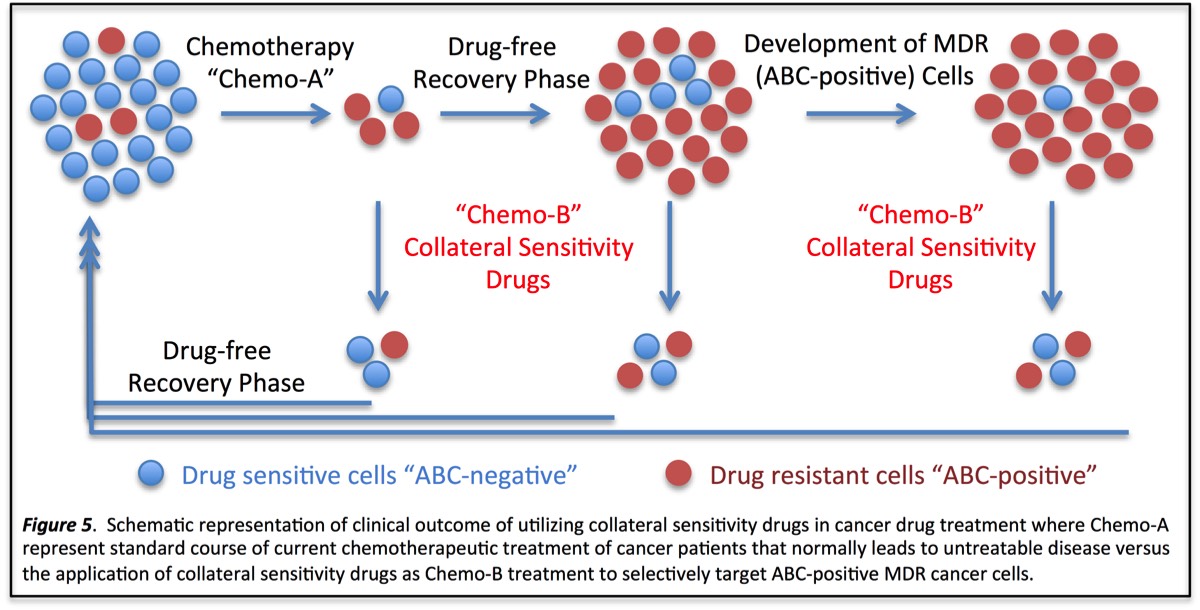
The goal of our research program on Collateral Sensitivity is to develop a clinical strategy and highly effective drugs that target ABCB1 over-expressing resistant tumours to prevent the failure of chemotherapy due to drug resistance. As suggested in the Figure 5, Collateral Sensitivity drugs can be given prior to chemotherapy (Chemo-A) to target ABCB1 over-expressing in tumour cells or post-treatment. Given that these drugs are not toxic to normal cells, it is envisioned that such drugs can be given to patients "pre and post-treatments". Together, this approach should lead to a significant advancement in cancer treatment and is likely lead to an increase in the overall survival of cancer patients.
Figure 4.
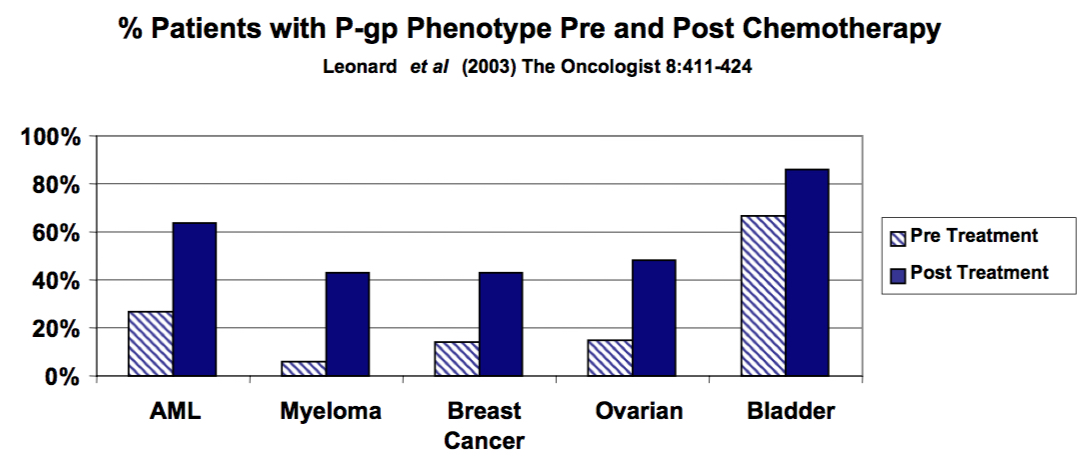
Figure 5.
Our Experimental Approaches
Project 1 - Identify drug combinations that act synergistically to induce collateral sensitivity in ABCB1 over expressing cancer cells and demonstrate their efficacy in vivo, using mouse models.
Project 2 - Map the entire collateral sensitivity pathway using CRISPR knock-out mutant cells to identify its key regulatory proteins, in addition to ABCB1 trigger.
Project 3 - Determine the role of ABCB1 post-translational modifications and protein interactions on its cellular trafficking, collateral sensitivity phenotype and drug efflux functions.
Peer-reviewed manuscripts on ABC-Mediated Collateral Sensitivity
P-glycoprotein mediates the collateral sensitivity of multidrug resistant cells to steroid hormones. Laberge RM, Ambadipudi R, Georges E. Biochem Biophys Res Commun. 2014 May 16;447(4):574-9.
A tamoxifen derivative, N,N-diethyl-2-[4-(phenylmethyl) phenoxy] ethanamine, selectively targets P-glycoprotein-positive multidrug resistant Chinese hamster cells. Georges E, Lian J, Laberge R. Biochem Pharmacol. 2014 Jul 15;90(2):107-14.
P-glycoprotein (ABCB1) modulates collateral sensitivity of a multidrug resistant cell line to verapamil. Laberge RM, Ambadipudi R, Georges E. Arch Biochem Biophys. 2009 Nov;491(1-2):53-60.
Modulation of GSH levels in ABCC1 expressing tumor cells triggers apoptosis through oxidative stress. Laberge RM, Karwatsky J, Lincoln MC, Leimanis ML, Georges E. Biochem Pharmacol. 2007 Jun 1;73(11):1727-37.
A mechanism for P-glycoprotein-mediated apoptosis as revealed by verapamil hypersensitivity. Karwatsky J, Lincoln MC, Georges E. Biochemistry. 2003 Oct 28;42(42):12163-73.